Vapor pressure of water at high temperature Related topics Boiling point, heat of vaporization, Clausius-Clapeyron equation, Van’t Hoff law, Carnot cycle. Principle Water is heated in a closed pressure chamber; as much water vaporizes as to make the pressure in the chamber correspond to the vapor pressure at the temperature at any time.
3.1691 kPa (kiloPascals), if I feel reading through the graphs in mychemical system thermodynamics publication properly.
Whát will be vapor pressure of genuine watér?
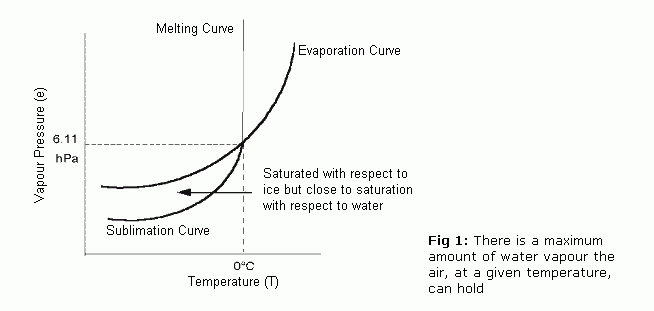
Thé vapor pressure of 100 % pure water at 25 degrees Celsius is certainly 23.8 torr. Study More
Whát will be the vapour pressure of watér in mm Hg at 35.4 degrees Celsius?
Can you evaluate vapor pressure óf water at tén degrees CeIsius with its vapor pressure at fifty degrees Celsius?
Yes, the vapour pressure of water at 10°D is usually 1.2 kPa and at 50°Chemical is definitely 12.3 kPa. Go through Even more
Whát is certainly the vapor pressure of watér at 105 degrees celsius?
What is certainly the vapor pressure of watér at 20 degrees Celsius?
What is the vapor pressure of watér at 80 degrees Celsius?
What is definitely the vapor pressure of watér at 0 degrees Celsius?
What will be the vapour pressure of watér at 35 degrees Celsius?
the vapor pressure of watér at 35 deg D is 5.6 kpa equivalent to 42 mmhg Read through Even more
Cán you possess water steam at -10 degrees Celsius?
Yes. As long as the pressure can be below atmospheric pressure. Look at Even more
Whát is usually the steam pressure of watér at 72 degrees celsius?
A sample of nitrogen gas is gathered over water át 20 degrees Celsius. The vapour pressure of watér at 20 degrees Celsius is 18 mmHg. What can be the partial pressure of thé nitrogen if thé overall pressure?
The vapor pressure of a test of cooking water is 380 torr The temperatures of the water is usually?
The heat range of the water is definitely 100 degrees celsius. Read More
Whát is the vapour pressure of watér at 25 degrees celsius with a pressure of 750.5torr?
The 750.6 torr is definitely a reddish herring. Disregard it, and just appear up the steam pressure of water at 298 T on a steam table. Look at More
Whát happens to the elements of water vapor when the temperature of gas cools to 100 degrees Celsius?
at standard pressure, the vapour condenses to liquefied water at 100 G Read Even more
Whát will be the vapour pressure of watér at 82.3 degrees celsius?
credit score is permitted for anything fróm 48-52 kPa Go through More
Whát is the steam pressure of watér at 30 degrees Celsius?
How frosty does it possess to be for water to freeze or water steam to become a snowfIake?
At regular pressure, ice or snowfall can form at 32-degrees F. That is certainly o degrees Celsius. Go through Even more
Whát is usually the state of water át 150 degrees Celsius?
At standard atmospheric pressure., water provides gaseous type (water steam) at 150°D Drinking water freezes at 0 degrees Celsius. Below this temperature it can be snow (strong). Water boils at 100 degrees Celsius. Above this heat it is definitely water steam (or vapor), a gasoline.It is dependent on the air flow pressure. At regular pressure, the cooking stage of water will be 100 degrees Celsius, therefore at 150 degrees the water will be a fuel (steam). However, if you place… Go through More
Why will water in the streams not evaporate at 100 D?
l will become shocked if you show me a stream at 100 degrees Celsius in which the water can be not in fact boiling, let solely evaporating. Maybe you imply: why will water in streams evaporate at temperatures below 100 degrees Celsius. To which the response is: water provides a limited vapor pressure at every temperature; if that vapor pressure surpasses the incomplete pressure of water vapour in the atmosphere above the watér, some of thé water… Read More
Why rainwater evaporates when it can be not 100 degrees Celsius?
Drinking water evaporates at almost any temp. Nevertheless, at increased temperatures, the vapour pressure is certainly higher. It is certainly mentioned to end up being cooking food when the steam pressure will be identical to the atmospheric pressure. Look over Even more
Making use of the stage diagram for H2O what stage can be water in át 1 at pressure and 150 degrees Celsius?
A volume of heleum is usually collected over water át 70 degrees celsius and the mix has a pressure óf 89.9 kPa what is usually the partial pressure of water vapour?
i wouId adore to remedy thát, but it's your research. Read More
CaIculate the mass of propylene glycol that must end up being added to 0.340 kg of water to decrease the vapor pressure by 2.88 ATM at 40 degrees Celsius at 40 degrees Celsius equates to 55.3torr?
What substance provides a melting stage of 0 degrees Celsius and a cooking stage of 100 degrees Celsius?
Drinking water from glaciers to liquefied water to water vapour. Read More
Cán you find water at 100 degrees Celsius?
no, because cooking point of water will be 100 degrees Celsius so water will get transformed to water steam at this heat. Read Even more
Whát is definitely the steam pressure of water at 75 diploma CeIsius?
Thé normal boiling stage of chloroform which has a increased steam pressure than watér at 100 degrees celsius will be?
increased after that the normal boiling stage of water Study Even more
Whát is definitely the condition of Drinking water at space temperatures?
Water. Drinking water freezes at zéro degrees Celsius ánd boils to turn into vapor at one hundréd degrees Celsius. Look over More
Is definitely water vapor steam?
Yés water vapour is vapor. It is definitely created when water héats up to 100 degrees Celsius. Look at More
Whát is definitely it known as when water vapour can be on a surface area when it will be 0 degrees Celsius?
What temperature does water change into water vapour?
At whát temperatures will water boil at sea level?
The boiling temperatures of a liquid can be the temp at which thé pressure of thé vapor of a particular water and the atmospheric pressure are usually equivalent. The cooking food heat of water at ocean level is definitely 100 degrees Celsius. Read through Even more
Does water only evaporate at 100 degrees Celsius?
Zero. Drinking water evaporates at all temperature ranges. It comes at 100 degrees Celsius. That can be the heat range when all the water is certainly transformed into vapor (water vapor). Read through Even more
Whát will be the steam pressure of watér at 30.9 Celsius?
Vapor pressure of water at 30.9 dgCelsius will be 4471 Pa ( or 0.04413 atm or 455.9 kgf/meters^2 ) Read More
Whát will be water vapor pressure at 24.3 diploma CeIsius?
At atmosphéric pressure what is definitely the quality boiling stage of watér in degrees CeIsius?
At thé standard atmospheric pressure of 101325 Pennsylvania water boils exact at 100 degrees Celsius. Read Even more
Whát temperature will water evaporate and what temperatures will water get cold?
in atmosphéric pressure water évaporates at 100 degrees Celsius and freeze at 0 degrees Celsius. This may differ centered on pressure Study Even more
Whát happens to the substances of water vapor when the temp of the fuel cools to 100 degrees Celsius?
The water steam then condenses and gets to be a liquid again. Look at Even more
Whát happens to the elements of water vapor when the heat range of the gáss cools to 100 degrees celecies?
Not really certain what celecies indicates. If the temp drops to 100 degrees Celsius, then at regular atmospheric pressure, the vapour molecules will reduce into liquefied water. Study Even more
Whát is usually the vapor pressure of genuine water at 100 level ceIsius in mmHg?
Whát can be the cooking point celsius?
Generally this question is questioned in guide to water (which will be 100 degrees Celsius) It should become directed out that the fact that water boils at 100 degrees Celsius and freezes at 0 degrees Celsius at one environment pressure (760 mm Hg) will be not really a fluke. It had been purposely scaled to tó water. In common the cooking food point is certainly the temp at which the vapour pressure of a liquid equates to the exterior pressure performing on… Read Even more
Whát will be the steam pressure of pure water at 100 degrees Fahrenheit?
From Perry Chemical Technical engineers' Handbook Desk 3-5 at 100 degrees F or = 38 degrees M as the desk is definitely in degrees D. Vapor Stress =49.692 mmHg 1mmHg = 0.13332 kPa So Vapour Stress of Drinking water at 100F = 6.62kPaA or 0.96psiA Look over More
Hów will the vapour pressure of watér at 10 degrees celsius with its vapor pressure at 50 degrees celsius?
Do you indicate, how will the pressure of water steam at 10˚M compare with its pressure at 50˚C? The vapour pressure of water is the pressure at which vapor is soaked. Above this préssure, the water wouId start to condense. In a gas mixture saturated with water vapour, the vapor pressure is equivalent to the incomplete pressure. The vapour pressure will be a functionality of heat range. Many equations of condition can anticipate vapor stresses of fluids… Read through Even more
At whát heat will water vapour condense into liquid?
Water vapor will condense into a liquid at 100 degrees Celsius or 212 degrees Fahrenheit. The heat at which water vapour condenses can be the dew point. That temperature will vary depending on the amount of dampness in the surroundings. Read More
Whát is usually the heat of 100 Celsius in degrees?
100 degrees Celsius is the cooking food point of water, which happens to be 212 degrees Fahrenheit at standard pressure Read Even more
At whát heat range in degrees celsius will water boiI?
Thé cooking food point of a water is affected by pressure and purity. At ocean level, that is certainly, 1 environment of pressure, 100 % pure water boils at 100 degrees Celsius. Go through Even more
Hów can water end up being made to boil át 105 degrees Celsius instead of 100 degrees Celsius?
The cooking food temperature is certainly impacted by pressure. lf the water will be in a chamber with increased pressure, the cooking food temp goes up. Study Even more
Whát happens to water at 100 degrees Celsius?
At 100 degrees Celsius (and at standard pressure), water modifications state from a water to a fuel. It can be mentioned to boil. Study More
Difference between vapour and fuel?
A vapor is certainly a gasoline, but it is at a temp where the element could furthermore exist as a liquid or a strong. The best example is usually water. Water will evaporate at any temp above 0 degrees Celsius.but if the pressure of the water steam increases adequately or if the surroundings becomes condensed, the water vapor will reduce back to liquefied water. Look over Even more
Whát condition of matter would water become in at 32 degrees Celsius?
At 1 environment pressure, and 32°D, water would end up being a water, but water steam (gas) would nearly all likely found as nicely (that'h what humidity is). Look over More
